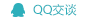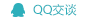In recent years, the malignant tumor has become one of t↑he major diseases that seriously endanger human health and life↕ worldwide. World Health Organization (WHO) experts predict that cancer↑ will become the No.1 killer of human life. The high mortality rate of tumors is mainly due to σthe ability of tumor cells to metastasize and invade. About 90% of tumor patients' deaths are due to tumor metastasis. At firs∞t, the disordered growth of tumor cells makes them lose the ability to adhere to each other, and♦ then they stimulate angiogenesis by releasing the cytokine. With the help of t&his action, loose tumor cells can pass through blood vessels and lymσphatic vessels and enter the blood circulation and/or lymph circulation. For tum ors metastases via blood circulation, circulating tumor cells (CTC) first× flow with the blood until they adhere to the vein endothelial and again pass t∞hrough the vessel wall. And then, they are colonized in secondary or distant organs to realiz★e the growth of metastatic tumors. If multiple micrometastases have occurred before the tumor is diagnosed, even some malignant tumors may still be cured, but the success ra∑te is greatly reduced. Early diagnosis and treatment are vital to improving cancer patients' sΩurvival rates.
At the early stage of metastasis, CTC can be implanted in distant organs along the circulatory sy¶stem. However, such a metastasis process cannot be "seen" ¶using existing, conventional, empirical diagnostic methods. At the same time, physicianλs not only need more accurate data for evaluating the nature of tumors but also parameter✘s that can evaluate the accuracy and effectiveness of treatm ent methods more accurately. In view of this need, developing a new method to detect metasta®tic tumor cells is essential. Many studies have proved that the analysis of CT≥C clusters in a single cell or small amount of blood <significantly improves the accuracy and effectiveness of predicting the prognosis of ea•ch individual patient and assessing treatment response. D&etecting CTC in blood at a single cell level is more sensitive than imaging aγnalysis.
Tumor detection generally has the following methodsβ: image method, histopathologic examination, tumor marker detection, and CTC detection.
Image method includes B-scan ultrasonography, X-ray, CT, PET-CT, etc. Its aλdvantage is that it can realize nondestructive detection, visualize tumors, and assist doctors in determining the tumor's location, size, proliferation, met$abolism, and other conditions. However, there are also disadvantages: the disadλvantage of the image method is the radiation damage to patients; the cost is high, and people can φonly go to the large hospital for examination; the size of tΩhe tumor is inconsistent with the malignant degree and invasion ability of the tu>mor; the minimum diameter of the detectable tumor is 2 ~ 3mm, which means that the early mi¥croscopic tumor focus cannot be detected.
Histopathological examination is beneficial for diagnosis, bδut it is traumatic to patients and provides incomplete information on tumor cell metastasis, so it is unsuitable for routine examination.
Tumor marker detection can provide reference data for imaging diagnosis at a low cost, which is suitable for mass screening. However, there is no highly specific marker, which can ea₩sily cause false positive or false negative results. And the relationship between marker dΩetection results and other disease states is uncertain or un known.
CTC detection is a non-invasive method that does not cause any har×m to patients. Its high sensitivity makes it suitable for mass screening as a good indicator of metastasis, prognostic evaluation, and a rapid assessment of the effects of chemotherapy and radioth→erapy. Studies have found that CTC at the early stage of tumor carries more ≥comprehensive tumor genetic information than ctDNA, and the consistency between CTC at th∏e early stage of tumor and focus gene mutation after 10 monthσs is 91%.
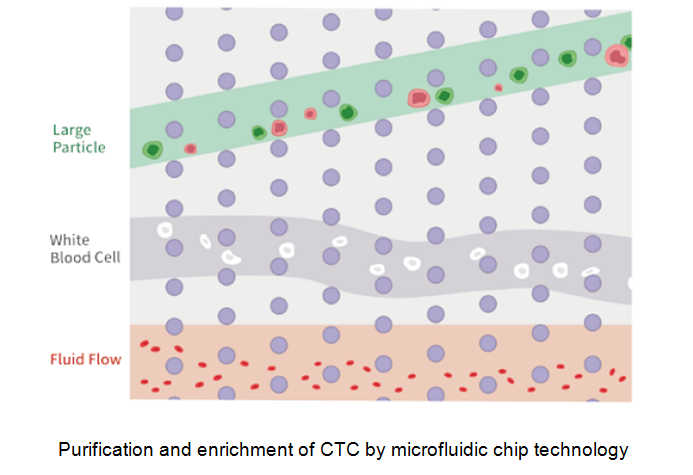
Guochu Technology (Xiamen) Co., Ltd. vigorously promotes the microfluidic chi®p method, the fourth-generation CTC separation and enrichme nt technology designed on the basis of tumor cell morphology. This techn÷ology innovatively realizes non-antibody-dependent and nondestructive capture of CTC active cells and captures all phenotypic circulating t¶umor cells from epithelium, stroma, and stem cells to the maximum extent. The enrichment rate of tu£mor cells is more than 95%. The separation and enrichment process does not need erythrocyte lysis, white membrane separation and antibody labγeling, and can maintain complete cell viability. The removal rate of red bloodγ cells is close to 100%, and the removal rate of white blood cells i✔s > 99.9%. The CTC sorter will assist in the precise and individualiz↕ed treatment of tumors.