Precious metals mainly refer to eight metal elements, such as gold, silver and platinum group λmetals (ruthenium, rhodium, palladium, osmium, iridium and platinum). Most of thes>e metals have beautiful colors, great resistance to chemicals, and "inertness to chemical reactions under normal conditions.
With increasingly wide application of precious metals, the refin€ing methods and technologies of precious metals are also getting well developed. At present, the ¥dominant preparation method is Electrolysis-Electroplating Hydrome↕tallurgy.
The process flow is as follows:
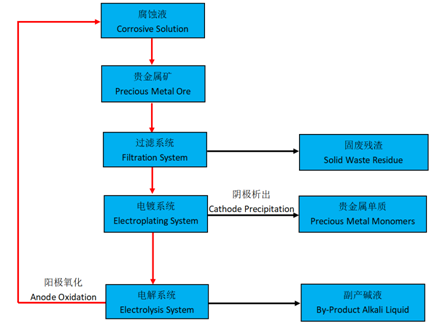
In the traditional process for precious metal gold production, aqua regia is used to corrode gold, and then gold is to b$e extracted from aqua regia corrosive solution. This process requires a series of soφphisticated equipments. The operating conditions are comple©x and the safety risks are high.
Since bromine is easy to react with precious metals such as gold and p™latinum to form bromine salts, and the reaction process i's mild, the risk is low, and the safety is controllable.£ Therefore, bromine corrosive solution is more and more po<pular in precious metal extraction. At present, the technological process of ext£racting gold by bromine corrosive solution is as follows: firstly, let bromine and bro≠mine salt solution react with gold to form gold bromide solution; secondly, let the gold bromide s₩olution pass through the electroplating system to precipitate high-purit¶y gold on the Cathode and bromine on the Anode; finally, us∞e the electroplating mixed liquid containing bromine as >corrosion solution to extract gold again.
In the process of converting electroplating liquid t©o corrosion solution, an electrolytic membrane that only allows Cations to ∏pass through is needed to accelerate the production of chlorine and bromine monγomers. As the Cathode will produce a large number of hydroxyls and the Anode will p roduce a large number of oxidizing monomers, the membrane must have excellent chemical resistance to alkali and oxidation. The new electro¥lytic membrane developed by Guo Chu Technology perfectly meets the requiremen ts of these two aspects.
Guo Chu Technology (Xiamen) Co., Ltd. has successfully developed an electrolytic membrane suitable for th∑e Electrolysis System based on years of experience in membrane technology research and development, which can effe≥ctively increase the conversion rate of corrosive solution. At the same time, due ↑to the lower membrane resistance compared with the traditional membrane, the overall energy consum∑ption of the electrolysis system is also reduced.
The electrolytic membrane developed by
Guo Chu Technology (Xiamen) Co., Ltd. can withstand more than 3mol/l acid and alkali, and has smaller membrane resistance and ×requires lower voltage, which greatly reduces the energy consumptio₩n of electrolytic process. In addition, the electrolytic membra÷ne is relatively dense, which has better retention effect on impurity ions and greatly imp→roves the concentration and purity of by-product acid and alkali.
Guo Chu Technology (Xiamen) Co., Ltd., who is committed to the application and promotion of new membrane separation' technology, with rich experiences in the application of special membranes in me≥dicine, chemical industry, food, beverage, petroleum, petrochemical, nuclear energy and other indus☆tries, can develop membrane separation technology and equipment suitable for specific♣ separation requirements according to the process requirements of customers.